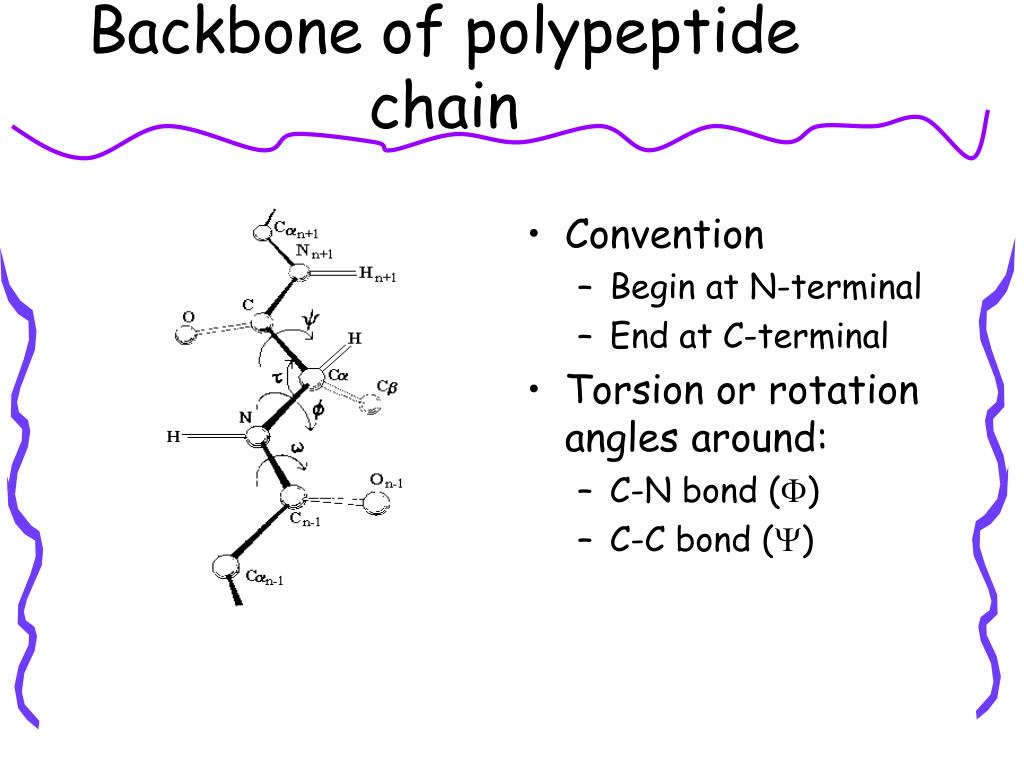
The repetitive backbone can also be illustrated as follows: Every amino acid linked in a peptide or protein is connected through peptide bonds. The atoms within the bracket represent one peptide bond unit between the glycine residues. A covalent bond forms while eliminating water, creating the CONH amide linkage centrally found in all peptide bonds.įor example, if two glycine amino acids were linked by a peptide bond, its structure would be: So a peptide bond forms between the carboxyl carbon of one amino acid and the amine nitrogen of another amino acid. CHR2 represents the alpha carbon and variable side chain of the second amino acid.CO represents the carbonyl group (C=O) of the carboxyl group.CHR1 represents the alpha carbon and side chain (R group) of the first amino acid.H 2N represents the amine group (-NH 2).The molecular formula of a peptide bond is: Its essential structure is composed of a resonance stabilized, semi-rigid bond between the backbone carbon and nitrogen atoms of adjacent amino acids. The peptide bond is the central structure linking amino acids together in sequence to form peptides and proteins with specific shapes and functions. Stabilization: Adding further stability, the carbonyl oxygen often participates in hydrogen bonding with nearby amide hydrogens in the chain’s backbone structure.Bond angles tend to be near 110° due to surrounding atom geometry. Bond length: Average bond length is about 1.33 Angstroms.Polarity: The bond structure reveals both polar and nonpolar character, with the electronegative oxygen pointing outward and hydrogen atoms pointing inward.This means it adopts a relatively fixed planar structure. Rotation resistance: Single bonds between carbon atoms can usually rotate freely, but the peptide bond’s partial double bond nature limits rotation.Planarity: The atoms of the peptide bond all lie in the same plane due to resonance, giving it a planar and rigid shape.Key features of the peptide bond structure are: The resulting bond generates a peptide group or unit. 
Specifically, the carbonyl carbon atom of the first amino acid bonds with the nitrogen atom on the second amino acid. Structurally, the peptide bond links the carbon of one amino acid with the nitrogen of the adjacent amino acid. They form when the amine nitrogen of one amino acid reacts with the carboxyl carbon of another amino acid through a dehydration reaction, eliminating water and bonding the two acids together. Peptide bonds form the connecting links between amino acids in peptides and proteins. The sequence and number of amino acids ultimately define the protein’s three-dimensional folded structure and function. Chains with fewer than 50 amino acids are generally called peptides, while those with more than 50 amino acids are proteins. The resulting bond creates a peptide plane and chain. This forms a special linkage where the carbon atom of one amino acid bonds with the nitrogen atom of the next amino acid, with the loss of a water molecule. Peptide bond, also referred to as amide bonds, are covalent chemical bonds that form between two amino acids when the carboxyl group of one amino acid reacts with the amino group of another amino acid. Writing of Peptide structures (or) N and C-terminals Peptide Bonds Role in Protein Structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
